
Concept Medical Inc., focused on vascular intervention drug delivery devices, has updated the progress of IMPRESSION (sIroliMus coated balloon angioPlasty versus plain balloon angioplasty in the tREatment of dialySis acceSs dysfunctION) randomised trial.
IMPRESSION is a prospective, multi-center, two-arm parallel group, randomised clinical trial to compare the efficacy of Sirolimus Coated Balloon versus Placebo Plain Balloon (control arm) in the treatment of dysfunctional (stenosed) fistula used for vascular access in hemodialysis. A total of 170 patients with End Stage Renal Disease (ESRD) are being randomised in 1:1 fashion (MagicTouch AVF: Placebo Plain Balloon).
The study’s primary objective is to determine if the use of Sirolimus Coated Balloon will result in an improvement of the primary circuit patency at six months when compared head-to-head with plain balloon angioplasty (POBA). The MagicTouch AVF Sirolimus Coated Balloon has already been granted the Breakthrough Device Designation by the U.S. Food and Drug Administration (FDA). Only ESRD patients with a matured dysfunctional arterio-venous fistula (AVF) that has been in use for at least one month prior to angioplasty will be enrolled.
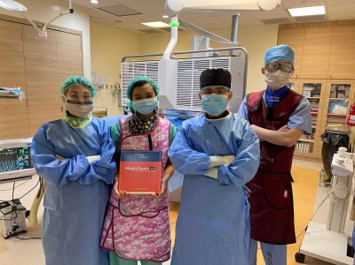
The study is being conducted at three sites in Singapore led by Associate Professor Chieh Suai Tan, Head and Senior Consultant, Department of Renal Medicine, Singapore General Hospital (SGH), as the principal investigator. Other site principal investigators include Associate Professor Edward Choke, Senior Consultant, Department of Surgery, Sengkang General Hospital, and Associate Professor Jackie Ho, Senior Consultant, Department of Cardiac, Thoracic and Vascular Surgery, National University Heart Centre, Singapore. The index patient was enrolled on 11th January 2021 and at the time of going to press, 37 patients have been enrolled.
SGH was chosen to lead this randomised controlled trial because of its prior experience using Sirolimus Coated Balloon in two investigator-initiated pilot studies. The studies had shown that patency rates with the use of MagicTouch AVF in thrombosed arterio-venous graft (AVG) and dysfunctional AVF were 65 per cent and 82.9 per cent at six months respectively.
An operational vascular access such as AVF or (AVG) is vital in ESRD patients for effective and long-term hemodialysis. Unfortunately, due to various reasons, the AVF or AVG may narrow (stenosis), resulting in poor flow of the vascular access, which affects hemodialysis. The functional life of an AVF is poor with as high as 50 per cent chance of failure after a median of three to seven years.
Singapore has one of the highest ESRD incident rates globally. POBA is the current standard treatment for patients with AVF stenosis. Despite the minimally invasive nature of POBA procedure, the long-term patency is poor, averaging 40 to 50 per cent at one year. This necessitates multiple procedures in order to maintain patency of the vascular access.
Paclitaxel Coated Balloon is also being used to treat dysfunctional AVF with mixed success. However, the safety concerns of Paclitaxel raised by the U.S. FDA and the issue of guidelines by the interventional societies have prompted doctors to seek a better alternative.
About MagicTouch AVF:
MagicTouch AVF is the first drug-coated balloon, that has been granted “Breakthrough Device Designation” from the U.S. Food and Drug Administration (FDA), for the treatment of Dysfunctional AVF and AVG. MagicTouch AVF is a CE approved and commercially marketed sirolimus coated balloon, developed using proprietary Nanolute Technology– the drug delivery technology platform of MagicTouch AVF balloon, is designed to deliver sub-micron particles of sirolimus designed to reach the deepest layers of the vessel walls.
About Concept Medical Inc (CMI):
CMI is headquartered in Tampa, Florida and has operational offices in The Netherlands, Singapore and Brazil and manufacturing units in India. CMI specializes in developing drug-delivery systems and has unique and patented technology platforms that can be deployed to deliver any drug / pharmaceutical agent across the luminal surfaces of blood vessels.

Disclaimer of Liability
The information, software, products, and services included in or available through CMI may include inaccuracies or typographical errors. CMI hereby disclaims all warranties and conditions with regard to this information, software, products, services and related graphics, including all implied warranties and conditions of merchantability, fitness for a particular purpose, workmanlike effort, title and non-infringement.
Indemnity Disclaimer
Users shall not transmit/publish or cause so to be done on the CMI website or any linked site, any material which is lascivious as appeals to the prurient interest or if its affect is such as to tend to deprave and corrupt persons who are likely to read, see or hear the matter contained or embodied in it. Users agree to indemnify and hold CMI, its subsidiaries, affiliates, officers and employees, harmless from any claim, demand, or damage, including reasonable attorneys’ fees, asserted by any third party due to or arising out of user’s use of or conduct on CMI services. CMI reserves the right to disclose any information about the use of the CMI services, including its contents, without user’s prior permission.
Copyrights
All content related to CMI corporate and business activities, as well as those of its associates included on CMI’s site as well as on associated sites, such as text, graphics, logos, button icons, images, audio clips and software, is the exclusive property of CMI and / or its associates, and is protected by applicable legislations concerning protection and preservation of intellectual property rights and international treaties relating thereto. The compilation (meaning the collection, arrangement and assembly) of all content on this site is also the exclusive property of CMI and/ or its associates and is protected by applicable laws as stated above. All software used on this site is the property CMI its associates or its software suppliers and are protected by copyright laws. The content and software on this site may be used as a shopping resource. Any other use, including the reproduction, modification, distribution, transmission, republication, display or performance, of the content on this site is strictly prohibited.
Non Transferable
User’s right to access CMI and related websites is not transferable. Any password, right or access given to User to obtain information or documents is not transferable and remains the exclusive property of CMI and or its associates as the case may be.
Applicable Law
This site is created and controlled by CMI in Surat, India; as such the laws of India shall apply; courts in Surat, India only shall have jurisdiction in respect of all the terms, conditions and disclaimers. CMI reserves the right to make changes to the site and the terms, conditions and disclaimers.
Intellectual Property Rights
“CMI respects intellectual property rights of third parties, and states that products will NOT be supplied to countries in which these could be in conflict with existing Intellectual Property Rights of a third party. CMI also maintains that the final responsibility lies exclusively with the buyer.”

Through your access and use of the Website, you accept, without limitation or qualification, the terms and conditions (“Terms and Conditions”) set forth below.
The Website contains information about products that may or may not be available in any particular country or region of the world, may be available under different trademarks in different countries and, where applicable, may be approved or cleared by a government regulatory body for sale or use with different indications and restrictions in different countries. However, the Website and its contents are designed to comply with India laws and regulations. Although accessible by others, it and its content are intended for access and use by Indian residents only.
The Website may contain from time to time information related to various health, medical and fitness conditions and their treatment. Such information is not meant to be a substitute for the advice provided by a professional health care provider. You should not use the information contained herein for diagnosing a health or fitness problem or disease. You should always consult a professional health care provider.
The Website contains information concerning CMI that may be useful to the Company’s customers and employees as well as the general public. In particular, CMI makes no representations or warranties as to the availability, accuracy or completeness of any information contained herein and expressly disclaims any obligation to update said information, except to the extent required by Food and Drug Administration regulations. CMI further assumes no liability or responsibility for any errors or omissions in the content of the Website.
The Website may contain financial and other information which is forward-looking and involves risks and uncertainties, including, without limitation, risks associated with the inherent uncertainty of pharmaceutical research, product development and commercialization, the impact of competitive products, patent, product liability and third-party reimbursement risks associated with the pharmaceutical industry, and the other risks and uncertainties detailed from time to time in CMI’s Annual Report.
Through your access and use of the Website, you accept, without limitation or qualification, the terms and conditions (“Terms and Conditions”) set forth below.
Terms and Conditions
You may freely browse the Website. You may freely download material therefrom. Any and all downloaded materials may be used by you for personal, non-commercial use. You may not, however, distribute, modify, transmit, reuse, repost or use any of such materials for public or commercial purposes, without the express written permission of CMI. All copyright and other proprietary notices contained in downloaded materials must be retained.
You should assume that all contents of the Website are copyrighted unless otherwise noted and may not be used except as provided herein and without the express written permission of CMI. Images of people or places displayed on the Website are either the property of, or used with permission by, CMI.
The use of these images by you is prohibited unless specifically permitted by these Terms and Conditions or by specific permission provided elsewhere on the Website. Unauthorized use of the images may violate copyright, trademark, privacy and/or publicity laws as well as communications regulations and statutes.
Your use of the Website is at your own risk. Neither CMI nor any other party involved in creating, producing or delivering the Website is liable for any direct, incidental, consequential, indirect or punitive damages arising out of your access to, or use of, or browsing in the Website. This includes damages to, or arising out of viruses that may infect, your computer equipment or other property. Without limiting the foregoing, everything on the Website is provided to you “AS IS” Without warranty of any kind, either expressed or implied, including, but not limited to, the implied warranties of merchantability, fitness for a particular purpose or non-infringement. Please note that some jurisdictions may not allow the exclusion of implied warranties, so some of the above exclusions may not apply to you. Check your local laws for any restrictions or limitations regarding the exclusion of implied warranties.
Any communication or material that you transmit to, or post, on the Website by electronic mail or otherwise, including any data, questions, comments, suggestions or the like, is, and will be treated as, non-confidential and non-proprietary information. CMI, or any of its affiliates, may use such communication or material for any purpose whatsoever, including, but not limited to, reproduction, disclosure, transmission, publication, broadcast and further posting. Further, CMI and its affiliates are free to use any ideas, concepts, know-how or techniques contained in any communication or material you send to the Website for any purpose whatsoever, including, but not limited to, developing, manufacturing and marketing products.
The trademarks, logos and service marks (collectively, the “Trademarks”) displayed on the Website are registered and unregistered trademarks of CMI and others. Nothing contained in this Website should be construed as granting, by implication, estoppel or otherwise, any license or right in and to the Trademarks without the express written permission of CMI or such third party. Your misuse of the Trademarks on the Website and in its contents, except as provided in these Terms and Conditions, is strictly prohibited. You are advised that CMI will aggressively enforce its intellectual property rights in the Website and its contents to the fullest extent of the law, including by seeking criminal sanctions.
CMI has not reviewed all of the Websites that may be linked to the Website and is not responsible for the content of any off-Website pages or any other Websites linked to the Website. Your linking to any other off-Website pages or other Websites is at your own risk. Once you link to another Website, please be sure to review the legal notices and privacy policy of the new Website. They may be different from this Website.
Although CMI may, from time to time, monitor or review the discussions, chats, postings, transmissions, bulletin boards and the like on the Website, CMI is under no obligation to do so, except to the extent required by Food and Drug Administration regulations, and CMI assumes no responsibility or liability arising from the content of the Website nor for any error, defamation, libel, slander, omission, falsehood, obscenity, pornography, profanity, danger or inaccuracy contained in any information within the Website.
You are prohibited from posting or transmitting any unlawful, threatening, libellous, defamatory, obscene, inflammatory, pornographic or profane material or any material that could constitute or encourage conduct that would be considered a criminal offense, give rise to civil liability or would otherwise violate the law. CMI will fully cooperate with any law enforcement authorities or court order requesting or directing CMI to disclose the identity of anyone posting any such information or materials.
You are prohibited from altering, damaging or defacing the Website or adding any unauthorized material to the Website. You are advised that CMI will aggressively protect the Website and will prosecute violators to the fullest extent of the law, including by seeking criminal sanctions.
CMI reserves the right to alter or delete material from the Website at any time, and CMI may, at any time, revise these Terms and Conditions and the other Policies set forth in this Website by updating this posting. You are bound by any such revisions and should, therefore, periodically visit this page to review the then current Terms and Conditions and Policies.
Privacy policy
We do not collect any personally identifiable information about you unless you provide it to us voluntarily. If you do not want your personally identifiable information to be collected, please do not submit it to us.
If you do provide us with personally identifiable information, such as your name, postal address, e-mail address, telephone number and other personal information, we may contact you, via e-mail or regular mail to provide you with information we believe may be of interest to you. At the time, you will be offered the opportunity to opt out of this service. We do not sell or transfer to others outside our company the information you send to us, except that your data may be transferred to an outside fulfilment company for processing and mailing purposes only. We consider your e-mail address and any personal information you give to be private and these data will be kept confidential within CMI and by any outside Fulfilment Company. Within CMI, data may be transferred to or accessed by authorized CMI personnel in other countries. At all times, personal data are treated confidentially.
Data that are not personal, such as questions, comments, ideas and suggestions, shall be deemed to be non-confidential, and CMI shall be free to disclose and use the same, without any obligation whatsoever to you, for any purpose and through any means.
In addition, we may store some information on your computer to improve the quality of your visit to our Website. In some cases, this information will help us to tailor the content to your interests, or help us to avoid asking for the same information on repeat visits to the Website. With most Internet browsers, you can erase the information, referred to as “cookies” from your computer’s hard drive, block all cookies or even receive a warning before a cookie is placed on your system. Refer to your browser instructions or help button to learn more about cookies.
We are very concerned about protecting the privacy of children. We do not intend to collect any personally identifiable information from children under 18 years old. If a child has provided us with personally identifiable information, the parent or guardian of that child should contact us.
If at any time you do not wish us to contact you or if you have questions concerning our privacy practices, please feel free to reach out to us.

We are committed to protect the privacy and security of your personal data. We advise you to carefully read this Privacy Notice (“Notice”) so that you are aware of how, where, and why we are using your personal data.
INFORMATION WE MAY PROCESS (“Personal Data”)
We may collect personal data on this website, including information we collect while performing business transactions and information we collect from other sources, as permitted by law.
We may collect your personal data including but not limited to
MEANS OF PROCESSING DATA
We may process your Personal Data through the following means:
COOKIES
Cookies are alphanumeric identifiers with small amount of data that is commonly used as an anonymous unique identifier. These are sent to your browser from the website that you visit and are stored on your computer’s hard drive. Please note, a cookie in no way gives us access to your computer, that is to say, cookies cannot access any other information on your computer.
Our Website uses these cookies to collect information and to improve our Service by making your interaction with us faster and more secure. They may be used for the purposes of managing your preferences, maintaining and optimizing security, marketing, communication, analytics, and research.
We majorly use below types of cookies:
If you do not want to receive cookies, most browsers allow you to control cookies through their setting preferences. However, if you do turn off cookies in your browser, you will not be able to fully experience some of the features our Website.
GROUNDS FOR LAWFUL PROCESSING OF PERSONAL DATA
When we process your Personal data, we do so primarily in connection with marketing, sale, and promotion of services which we provide or which are procured using our services and to improve and enhance the range of those services and offerings. We may process your personal data for recruitment, communication, sales and marketing, registration and subscriptions purposes, where processing is necessary for the purpose of pursuing our legitimate interests.
USE OF PERSONAL DATA
We generally use the information to establish and enhance our relationship with our users for the following purposes:
We keep your personal data only for the duration of legitimate business requirements or as required by law.
TRANSFERRING YOUR PERSONAL DATA
As a global business, we may transfer your personal data to servers, entities and partners or other trusted third parties based in other countries so that they may process personal data on our behalf. By using our Website or otherwise providing us with your personal data, you agree to us doing so in accordance with the terms of this Privacy Notice and applicable data protection laws and regulations. We will take all steps reasonably necessary to ensure that your data is treated securely and in accordance our data protection policy, which is in line with applicable data protection legislation. We may also transfer your data to third party service providers and web analytics tool providers. CMI may permit certain third party companies to help CMI tailor the advertising that it thinks may be of interest to you, based on your use of our Website.
CHILDREN’S PRIVACY
We understand the importance of taking extra precautions to protect the privacy and safety of children using our Website. The Services are not intended for users under the age of 16. We do not knowingly collect any Personal data from children under 16 or market to or solicit information from anyone under the age of 16. If we become aware that a person submitting Personal data is under 16, we will delete all the information as soon as possible unless it is with the consent and involvement of a parent or guardian. If you believe we might have any information from or about a child under 16, please contact us at privacy@conceptmedicals.com.
SECURITY: HOW SECURE IS YOUR INFORMATION WITH US?
We have implemented appropriate physical, electronic, procedural and managerial procedures to safeguard your information against loss, unauthorized access, misuse or modification. We require any third parties processing your information to do the implement the same levels of protection with respect to your data. We strive to use commercially acceptable means for protecting your information.
CHANGES TO THE NOTICE
This Notice is effective as of {date}*. We reserve the right to update or change this Notice at any time, and we will provide you with the updated Notice when we make any substantial updates at the earliest either through email or by providing a prominent notice of change on the Website. You should check the Notice periodically. Your continued use of the Website after we post any modifications to the Notice on this page will constitute your acknowledgment of the modifications and your consent to abide and be bound by the modified Notice.
THIRD PARTY WEBSITES
From time to time we may provide links to the websites of other organisations, these links are provided for your information only. We have no control over the contents of those sites or resources, and accept no responsibility for them or for any loss or damage that may arise from your use of them.
Your rights and your personal data
We will respect your legal rights in relation to your data. Below are the rights granted to individuals located in the General Data Protection Regulation, and CMI is committed towards protection of the same.
We retain personal information for as long as we reasonably require it for legal or business purposes. In determining data retention periods, CMI takes into consideration local laws, contractual obligations, and the expectations and requirements of our customers. When we no longer need personal information, we securely delete or destroy it.